5-Year-Old Died 4 Days After Pfizer Shot, CDC VAERS Data Show. Another of 11yo Deceased Earlier
by Fabio Giuseppe Carlo Carisio
All Italian links inside the article can be read in English with simultaneous machine translation
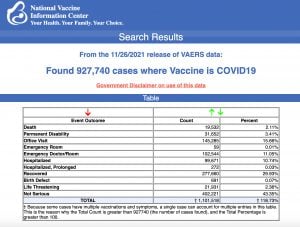
The Centers for Disease Control and Prevention today released new data showing a total of 927,740 reports of adverse events following COVID vaccines were submitted between Dec. 14, 2020, and Nov. 26, 2021, to the Vaccine Adverse Event Reporting System (VAERS). VAERS is the primary government-funded system for reporting adverse vaccine reactions in the U.S.
The data included a total of 19,532 reports of deaths — an increase of 283 over the previous week — and 146,720 reports of serious injuries, including deaths, during the same time period — up 3,325 compared with the previous week.
«Every Friday, VAERS publishes vaccine injury reports received as of a specified date. Reports submitted to VAERS require further investigation before a causal relationship can be confirmed. Historically, VAERS has been shown to report only 1% of actual vaccine adverse events» according to Megan Redshaw, journalist of The Defender, the website of Children’s Health Defense association led by Robert F. Kennedy jr.
«U.S. VAERS data from Dec. 14, 2020, to Nov. 26, 2021 for 5- to 11-year-olds show: 2,586 adverse events, including 34 rated as serious and 2 reported deaths. One death occurred in an 11-year-old girl from Georgia vaccinated Sept. 14, prior to the authorization of Pfizer’s COVID vaccine in the 5 to 11 age group. The second death (VAERS I.D. 1890705) occurred in a 5-year-old girl who died four days after receiving her first dose of Pfizer. 1,581 adverse events have been reported in the 5 to 11 age group since Nov. 1».
Among the 34 serious cases reported among children between 5 and 11 years there is also that of «a 5-year-old girl, in Florida, hospitalized with ataxia (lack of motor control caused by cerebellum problems) and high fever 3 days later having taken the Pfizer vaccine» as highlighted by the blog brezzadessenza.wordpress.com which also reports the number and details of the VAERS (Vaccine Adverse Event Reporting System) practice.
As detailed in the VAERS (Vaccine Adverse Event Reporting System) file, the 5-year-old girl deceased in Iowa had no birth defects and no known allergies. He received Pfizer-Biontech’s experimental Comirnaty gene serum on November 18 after emergency use clearance from the Food and Drug Administration and a recommendation from the Centers of Diseases Control and Prevention (CDC) committee voted amid a scandal for the funding received from a doctor on the evaluation panel from Pfizer partner GlaxoSmithKline, as revealed by Gospa News.

The little girl underwent a recent hospitalization for rhino/enterovirus, mycoplasma pneumoniae ab+ for acute respiratory failure. But she was nevertheless subjected to the administration of Pfizer with the consent of the doctors.
Due to the patient’s complex clinical history (who in the past had needed a transfusion which forced her to use a CPAP for night breathing) «provider asked that patient be monitored overnight after administration of COVID vaccine. Patient was moved from PICU to general peds floor due to improvement in condition on Thursday. Vaccine was administered that evening. Patient did well. Remained on room air. Was discharged home on Saturday. On Monday morning, father checked on patient and she was found pulseless and not breathing. It is unclear whether or not patient was placed on home CPAP during the night. EMS called. Patient arrived to ED as a CPR in progress. Patient presented with a pH of <6. Last known well 9pm the evening prior. Patient expired at 11/22/21 at 11:05 CST» we read in the VAERS file.
From the description of the clinical events it might appear that death was related to previous respiratory disease but complications presented according to standardized MedDRA queries (SMQs), which are tools developed to facilitate retrieval of MedDRA encoded data as a first step in investigating medical problems. drug safety in pharmacovigilance and clinical development, are those repeatedly reported among suspected adverse reactions to messenger RNA-based vaccines.
«Anaphylactic reaction (broad), Lactic acidosis (broad), Shock-associated circulatory or cardiac conditions (excl torsade de pointes) (narrow), Acute central respiratory depression (narrow), Guillain-Barre syndrome (broad), Hypersensitivity (broad), Respiratory failure (narrow), Infective pneumonia (broad)» we read in the clinical file of deceased child.
It will be up to a medical examiner, if an autopsy is required, to ascertain the causal link between death and these suspected side effects even though it is well known that anaphylaxis, Guillain-Barre syndrome as well as heart attacks are among the most common.
Especially cardiac inflammations (myocarditis or pericarditis) are widespread among young people and for this reason Pfizer itself, in its report to the FDA to obtain the emergency use of Comirnaty, had warned the advisory committee that it had not collected sufficient clinical data to «detect any potential risks of myocarditis associated with vaccination».
Despite this, first the FDA and CDC in the USA, then the European Medicines Agency (EMA) in the countries of the European Union and finally the AIFA (Italian Medicines Agency) have extended the use of experimental gene sera first to over 12 and, in recent days, to over 5 years old.
EMA and AIFA in their official statements have even concealed the risks from myocarditis although Pfizer itself had elaborated a statistic even predicting the number of hypothetical cases: «The estimated numbers of prevented COVID-19 cases and associated hospitalizations for 1 million vaccinated children 5 to <12 years of age during the 120 days post-vaccination are ~33,600 and 170, respectively; in contrast, 21 post-vaccination myocarditis cases would be expected. Looking separately at males and females, 43 myocarditis cases would be expected per 1 million fully vaccinated males and 4 cases would be expected per 1 million fully vaccinated females» wrote in its dossier (link below in the sources) the American multinational that with the German BioNtech produced Comirnaty, the only drug for – presumed – immunization to Covid authorized for minors.
The premature deaths of the Iowa girl and the 11-year-old from Georgia confirm, if ever they were needed, the high danger of these vaccines beyond Big Pharma’s scientific considerations on the risk-benefit ratio, although, according to what is admitted by Italian AIFA, the serious consequences of Covid-19 in children are practically non-existent.
«The data available in the reports of the ISS (Health Superior Institute of Rome) show in recent weeks a clear increase in the number of infections in the population of 5-11 years of age, where the greatest increase in absolute compared to any other age group. This observation can obviously be explained by the fact that children aged <12 years have not been vaccinated up to now. The aforementioned data also show that in the infected of this age group the hospitalization rate is approximately 6 / 1,000, while that of hospitalization in intensive care is approximately 1.4 / 10,000. A recent ECDC1 report also shows that most of the 5-11 year old children hospitalized for COVID did not have any risk factors» is what the AIFA Technical Scientific Commission wrote in the opinion with which it authorized the emergency use of Comirnaty also for children, while some doctors funded by Big Pharma are already pushing to extend it to newborns as well.
Precisely the lack of data on the potential risks of myocarditis and other adverse reactions in children reveals a terrible circumstance: even the youngest have become guinea pigs in a global experiments, as admitted by a German geneticist, that is really becoming almost similar to the crimes of Nazi doctors condemned by the Nuremberg Code.
MAIN SOURCES
https://www.gospanews.net/en/2021/08/24/british-medical-journal-contested-fda-for-political-decision-on-full-approval-of-pfizer-vaccine-without-public-discussion-of-data/




Hi there to all, how is the whole thing, I think every one is getting more from this web
site, and your views are fastidious designed for
new visitors.