EXCLUSIVE: Pfizer Paper confirms Genic Experiments on 5yo Kids. “Myocarditis Risks by Vaccines, Little Data… We”ll Study After FDA Authorization”

by Fabio Giuseppe Carlo Carisio
When about two weeks ago in the US the Food and Drug Administration authorized the emergency use of Pfizer-Biontech’s Covid Comirnaty vaccine also for children aged 5 to 11 (previously it had already been approved for adolescents between 12 and 15) the mainstream media have clung to that 90.7% effectiveness to extol the goodness of the decision.
5-Year-Old Died 4 Days After Pfizer Shot, CDC VAERS Data Show. Another of 11yo Deceased Earlier
Those of counter-information, on the other hand, including the Italian newspaper Il Tempo, focused on the opinions of skeptical doctors of the FDA Advisory Committee but almost no one went to read the official document provided by Pfizer inc – the American multinational that produces the genic serum with the German Biontech but is also a partner of the London-based GSK, another famous Big Pharma of vaccines – in order to obtain that authorization (Emergency Use Authorization).
Taiwan suspends 2nd doses of Pfizer for Teens over Health Risk
In the 82 pages of the dossier we found there is a truly explosive statement. In fact, Pfizer itself admits that a real genic experiment will be conducted on children for 5 years since the data of two studies (one conducted in the US and the other in Israel) involved about 3 thousand children.
Actually a very small sample, not sufficient to record possible serious adverse events such as the feared inflammation of the heart (myocarditis of the internal muscle tissue or pericarditis of the external lining) so widespread among adolescents as to induce Taiwan to suspend the second dose of Pfizer for young people and some Scandinavian countries to stop the inoculation of Moderna’s messenger RNA vaccine, technically similar to that of Pfizer, for the same reason.

«The number of participants in the current clinical development program is too small to detect any potential risks of myocarditis associated with vaccination. Long-term safety of COVID-19 vaccine in participants 5 to <12 years of age will be studied in 5 post-authorization safety studies, including a 5-year follow-up study to evaluate long term sequelae of post-vaccination myocarditis/pericarditis» Pfizer writes on page 11 of its Oct. 26 report called “Briefing Document” for the Vaccines and Related Biological Products Advisory Committee (VRBPAC).
The meaning may well be understood even by a 5-year-old child. FDA gives us its authorization for emergency use even if data on the most dangerous adverse reactions are missing, we will continue the studies: obviously thanks to the vaccination campaigns launched in the United States of America with the consent of the Centers of Diseases Control and Prevention, ended up in the eye of the storm over the scandalous conflict of interest between a doctor on the CDC Approval Committee and Pfizer’s Big Pharma GSK partner.
Inside FDA Committee the alarm about the lack of data for myocardial risks was confirmed by Leslie Ball, an FDA medical officer, who reiterated this concept, arguing that the studies were not large enough to necessarily collect an uncommon side effect such as myocarditis
Despite this, the FDA had approved the use of Comirnaty for children aged 5 and over as previously announced by the American president Joseph Biden, the great director of the mass immunization plan, in turn in macroscopic conflict of interest in relation to approximately $ 2.2 million received by Pfizer for the presidential election campaign of the Democratic Party.
In light of what has been discovered, it is not surprising that the political and health authorities of Vienna have already started the administration of Pfizer-Biontech’s experimental genetic serum also to 5-year-old children, despite the fact that the clinical data have not yet been examined by the European Medicines Agency for the granting of emergency commercial use. Probably the Austrian data could be used by the American pharmaceutical multinational to increase the volume of statistics on the risks of adverse reactions for children.
In its “Overall risk-benefit conclusions”, again on page 11, Pfizer reiterated the reasons for the need for experimental genic sera, although – as evidenced by a geneticist from Cologne – they can also cause the modification of human DNA.
«COVID-19 continues to be a serious and potentially fatal or life-threatening infection for children and there is a significant unmet medical need in the 5 to <12 years of age population. Two primary doses of the 10 μg BNT162b2 vaccine given 3 weeks apart in 5 to <12 years of age have shown a favorable safety and tolerability profile, robust immune responses against all variants of concern and high VE against symptomatic COVID-19 in a period where the delta variant was predominant» Big Pharma doctors wrote.
«Israeli safety surveillance databases suggest that incidence rates of rare post-vaccination myocarditis peaks in individuals 16 to 19 years of age males and declines in adolescents 12 to 15 years of age. In addition, the dose for children 5 to <12 years of age is 1/3 of the dose given to older vaccinees (10 μg vs. 30 μg). Based on this information, it is reasonable to predict that post-vaccine myocarditis rates are likely to be even lower in 5 to <12 years of age than those observed in adolescents 12 to 15 years of age».
The adverb “likely” is the clearest confirmation of a genetic experiment in progress.
«Given post-authorization experience and assuming 90% efficacy as shown in the descriptive clinical study, the estimated number of COVID-19 cases and associated hospitalizations prevented over 120 days per million of fully vaccinated children 5 to <12 years of age is ~33,600 and 170, respectively. In contrast, the number of post-vaccination myocarditis cases (including myocarditis, pericarditis, and myopericarditis) expected in the same period of time per million second doses is 21 (assuming that children 5 to <12 years of age experience the same rates of post-vaccination myocarditis/pericarditis as adolescents 12 to 15 years of age in the US)» adds Pfizer by publishing in section 4 a not very reassuring graph on the incidence of heart inflammation.
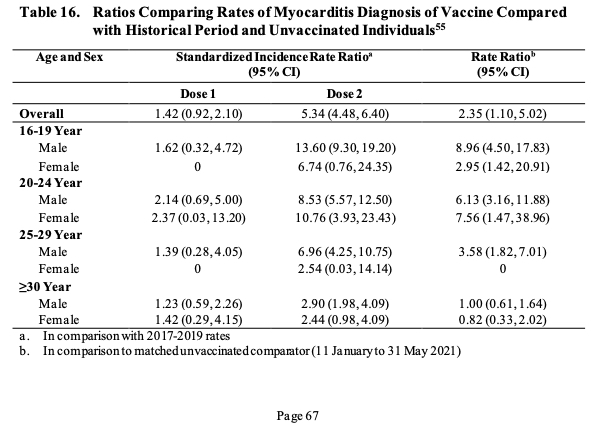
«We therefore may expect substantially fewer post-vaccination myocarditis cases among 5 to <12 of age males and females than COVID-19-associated hospitalizations given current age-specific COVID-19 cases and hospitalization rate estimates. Prevention of potential long-term sequelae of COVID-19 illness as well as other societal impacts would further increase the public health benefit of vaccination. Given all the above, the benefits of the Pfizer-BioNTech COVID-19 Vaccine to prevent COVID-19 given as a two-dose, 10 μg dose level, primary series in children 5 to <12 years of age outweigh the known or potential risks» is the conclusion of the Comirnaty manufacturing company.
But precisely in relation to the alleged benefits, some members of the FDA Committee had expressed skepticism. Dr. Ball had stated that in the FDA’s analysis, the agency was unable to determine if there was an impact of vaccination on the “spread” of the virus (in short, if and how the vaccine blocks infections).
Boys more at risk from Pfizer jab side-effect than Covid, suggests study
James Hildreth, a member of the FDA advisory committee and also president of Meharry Medical College in Nashville, Tennessee, ultimately voted to authorize the vaccine, but said it was a “challenging decision”. Hildreth said he was surprised to learn data from the CDC that up to 40% of children in this age group have so far been infected with the coronavirus, which he says could mean that up to 30 million children may already have some form of immunity in the United States. “It seems to me somehow that we are vaccinating children to protect adults. It should be the other way around,” Hildreth said.
Eric Rubin, a member of the FDA Advisory Commission, and professor of immunology at Harvard T. H. Chan School of Public Health and Brigham and Women’s Hospital, also admitted that “we decided to vote him with a heavy conscience,” according to Time. While Michael Kurilla, who heads a division within the National Institutes of Health, was the only member not to vote in favor of pediatric vaccinations. He abstained from voting, while all the other 17 members voted yes.
«Kurilla expressed concern that Pfizer-BioNTech has not provided detailed data on what happens to the antibodies to the virus that children generate after they are vaccinated, and whether these decrease as they do in adults. He was also concerned about the possibility of vaccinating children who may have already been naturally infected with the virus and may not need further protection from an injection» the Italian newspaper Il Tempo reported.
It is important to understand what the real risks are for children contracting COVID, and to do so we can quote an excerpt from the official British Vaccine Board (JCVI) paper: “Symptoms that are observed in children and young people, they are typically mild and little different from other mild respiratory viral infections that circulate each year. Children recover quickly from these infections and, according to many studies, develop robust broad-spectrum immunity”.
MAIN SOURCES




3 pensieri su “EXCLUSIVE: Pfizer Paper confirms Genic Experiments on 5yo Kids. “Myocarditis Risks by Vaccines, Little Data… We”ll Study After FDA Authorization””